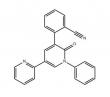
FDA Rationale for Recognition Decision: Ceftaroline fosamil
FDA has reevaluated the rationale document titled, “Ceftaroline Breakpoints for Staphylococcus aureus,” submitted by the Clinical and Laboratory Standards Institute (CLSI) to FDA public docket # FDA-2017-N-5925-020 in November 2019. The initial review was …